Yan-Jia FANG, Meng-Hua ZHOU, Xiao-Fei GAO, Hua GU and Yan-Ai MEI1
Institute of Brain Science, School of Life Sciences and State Key Laboratory of Medical Neurobiology, Fudan University, Shanghai 200433, China
Biochem. J. (2011) 438, 203–215 (Printed in Great Britain) doi:10.1042/BJ20110569
AA (arachidonic acid), which possesses both neurotoxic and neurotrophic activities, has been implicated as a messenger
in both physiological and pathophysiological processes. In the present study, we investigated the effects of both extracellular and
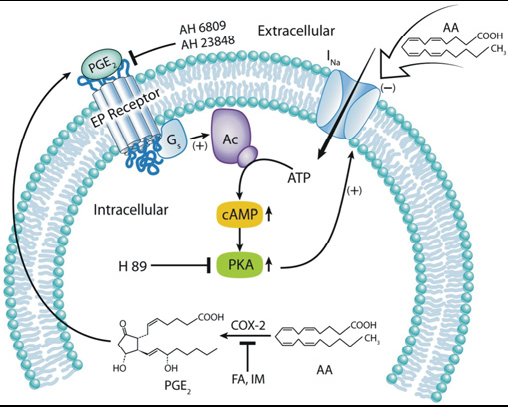
intracellular application of AA on the activity of NaV (voltagegated Na+ channels) in rat cerebellar GCs (granule cells). The extracellular application of AA inhibited the resultant INa (NaV current), wherein the current–voltage curve shifted to a negative
voltage direction. Because this effect could be reproduced by treating the GCs with ETYA (eicosa-5,8,11,14-tetraynoicacid) or a membrane-impermeable analogue of AA, AA-CoA (arachidonoyl coenzyme A), we inferred that AA itself exerted the observed modulatory effects on INa. In contrast, intracellular AA significantly augmented the elicited INa peak when the
same protocol that was used for extracellular AA was followed. The observed INa increase that was induced by intracellular AA was mimicked by the AA cyclo-oxygenase metabolite PGE2 (prostaglandin E2), but not by ETYA. Furthermore, cyclooxygenase inhibitors decreased INa and quenched AA-induced channel activation, indicating that the effect of intracellularAAon NaV was possibly mediated through AA metabolites. In addition, the PGE2-induced activation of INa was mimicked by cAMP and quenched by a PKA (protein kinase A) inhibitor, a Gs inhibitor and EP (E-series of prostaglandin) receptor antagonists. The results of the present study suggest that extracellular AA modulates NaV channel activity in rat cerebellar GCs without metabolic
conversion, whereas intracellular AA augments the INa by PGE2- mediated activation of cAMP/PKA pathways. These observations may explain the dual character of AA in neuronal pathogenesis.

